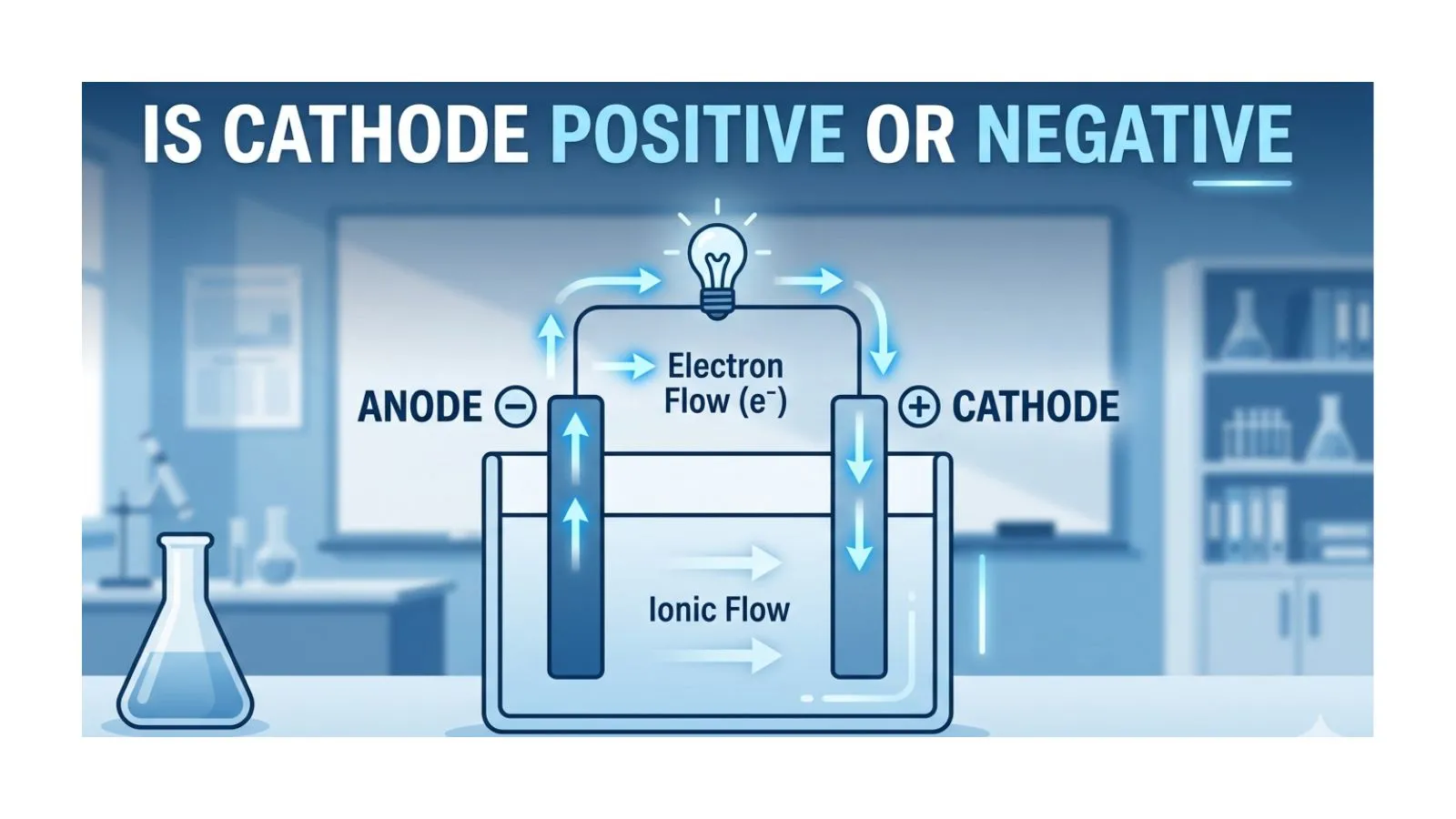
Many students and beginners in physics and electronics search the phrase “is cathode positive or negative” because this concept often feels confusing at first. The confusion happens because the answer is not always the same.
In some cases, the cathode is negative, and in other cases, it is positive. This depends on the type of device being used, such as a battery, diode, or electrolytic cell.
|See also Shit or get off the pot
People also get mixed up because textbooks sometimes explain the cathode in different ways. One definition says the cathode is where reduction happens.
Another definition says it is the negative terminal. Both are correct, but only in specific contexts.This article will clear all confusion in very simple words.
You will learn the quick answer, the origin of the term, differences in usage, and real-life examples. We will also show common mistakes students make and how to avoid them.
By the end, you will clearly understand when the cathode is positive and when it is negative without any confusion.
Is Cathode Positive or Negative – Quick Answer
The cathode can be positive or negative, depending on the system:
- In a galvanic (voltaic) cell (battery): Cathode is positive
- In an electrolytic cell: Cathode is negative
Simple rule:
- Cathode = site of reduction (gain of electrons)
- The charge depends on whether the device produces or uses electricity
Example:
- Battery → Cathode = + terminal
- Electrolysis of water → Cathode = – terminal
The Origin of Cathode
The word cathode comes from Greek:
- “kata” = down
- “hodos” = way
It was first used in early electricity studies to describe the path of current flow. Scientists used it to define electrodes in vacuum tubes and chemical cells. Over time, the meaning stayed the same (reduction site), but the charge changed based on the system.
British English vs American English Spelling
The word cathode is spelled the same in both British and American English.
There is no spelling difference, but usage explanation can differ in textbooks.
Comparison Table
| Term Context | British English | American English |
|---|---|---|
| Cathode spelling | Cathode | Cathode |
| Explanation style | More theory-based | More practical-based |
| Teaching focus | Electrochemistry detail | Circuit application |
Which Spelling Should You Use?
Since there is no spelling difference, you should always use “cathode” in all regions:
- USA → Standard scientific term
- UK/Commonwealth → Same term used in physics and chemistry
- Global use → Universally accepted word
Focus more on correct meaning, not spelling.
Common Mistakes with Cathode
Students often make these mistakes:
- ❌ Thinking cathode is always negative
- ❌ Mixing up cathode and anode
- ❌ Forgetting it depends on the system
- ❌ Not linking it with reduction reaction
Correct understanding:
- Cathode = reduction always
- Charge = depends on device type
Cathode in Everyday Examples
Email / Study Notes Example:
“The cathode is positive in a battery but negative in electrolysis.”
News / Science Example:
“Researchers improved battery efficiency by optimizing cathode materials.”
Social Media Example:
“Still confused about cathode vs anode 😅 it depends on the system!”
Formal Writing Example:
“In electrochemical cells, the cathode is defined as the electrode where reduction occurs.”
Cathode – Google Trends & Usage Data
The keyword “is cathode positive or negative” is commonly searched by:
- Students (high school & college)
- Exam preparation users
- Engineering beginners
It trends more in:
- South Asia (India, Pakistan)
- USA (education searches)
- UK (exam revision searches)
Search interest increases during:
- Exam seasons
- Physics revision periods
Comparison Table – Cathode Variations
| Situation | Cathode Charge | Reason |
|---|---|---|
| Battery (galvanic cell) | Positive (+) | Gains electrons |
| Electrolysis (electrolytic cell) | Negative (–) | Connected to external power |
| Vacuum tube | Negative | Electron emission |
| General rule | Neutral definition | Always reduction site |
FAQs:
What is a cathode and anode?
Cathode is the electrode where reduction happens, and anode is where oxidation happens.
Is cathode positive or negative in GCSE?
In GCSE chemistry, cathode is negative in electrolysis and positive in batteries.
Is cathode or _?
Cathode is the electrode that gains electrons (reduction site).
Is cathode higher than anode?
No, cathode is not “higher”; it only refers to reaction type, not position.
Is a cathode negative?
Not always. It is negative in electrolysis but positive in a battery.
Is anode left or right?
There is no fixed left or right; it depends on the diagram or setup.
Conclusion (150–200 words)
Understanding whether the cathode is positive or negative is simple once you know the rule. The most important point is that the cathode is always the site of reduction, meaning it always gains electrons.
However, its electrical charge depends on the system you are studying. In a battery or galvanic cell, the cathode is positive because it receives electrons naturally during chemical reactions.
In electrolysis, the cathode becomes negative because an external power source pushes electrons into it.Many students get confused because they try to memorize a single answer.
Instead, it is better to understand the concept. Once you remember that “cathode = reduction,” the rest becomes easy. You can then quickly figure out whether it is positive or negative based on the device.
This concept is very important in physics, chemistry, and electronics. It is also widely asked in exams and interviews. By learning it properly, you will avoid common mistakes and build a strong foundation in electrochemistry. Always focus on understanding, not memorizing, and you will never be confused again.